Sodium chlorate
Agent Name
Sodium chlorate
CAS Number
7775-09-9
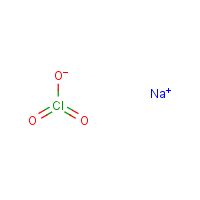
Formula
Cl-Na.-O3
Major Category
Other Classes
Synonyms
Agrosan; Asex; Atlacide; B-Herbatox; Chlorate de sodium [ISO-French]; Chlorate salt of sodium; Chloric acid, sodium salt; Chlorsaure [German]; De-Fol-Ate; Defol; Dervan; Desolet; Drexel defol; Drop-Leaf; Evau-Super; Fall; Grain sorghum harvest-aid; Granex O; Harvest-aid; Hibar C; KM; Kusa-tohru; Kusatol; Leafex 2; Leafex 3; Natrium chloraat [Dutch]; Natrium chlorat [German]; Natriumchloraat [Dutch]; Natriumchlorat [German]; Ortho C-1 defoliant & weed killer; Ortho-C-1-Defoliant; Oxycil; Rasikal; Shed-A-Leaf "L"; Shed-A-leaf; Soda chlorate; Sodakem; Sodio (clorato di) [Italian]; Sodium (chlorate de) [French]; Sodium chlorate [ISO]; Sodium(chlorate de) [French]; Travex; Tumbleaf; Tumbleleaf; United Chemical Defoliant No. 1; VAL-DROP; Weed Killer; [ChemIDplus] UN1495; UN2428 (aqeous solution)
Category
Other Inorganic Compounds
Description
Colorless or white odorless solid; [Merck Index] Slightly hygroscopic; [CHEMINFO] Colorless or light yellow granules; [MSDSonline]
Sources/Uses
Used as oxidizing agent and bleach for wood and paper pulps; in ore processing (uranium and vanadium); as herbicide, defoliant, desiccant, semi-permanent soil sterilant, and harvest aid; in matches, explosives, flares, and pyrotechnics; to recover bromine from natural brines; for leather tanning and finishing; as textile mordant and bleaching agent; to make perchlorates, potassium chlorate, chloric acid, and chlorine dioxide; as oxidizer in making dyes, rocket propellants, and pharmaceuticals; in toothpastes and mouthwashes; as a sweetening agent in oil refineries; [HSDB] Used in metal etching and oxygen generation in rescue breathing apparatus; [CHEMINFO]
Comments
A strong oxidant and fire hazard on contact with organic substances; [Hawley] A fire and explosion hazard on contact with combustible and reducing materials; A skin, eye, and respiratory tract irritant; Can induce methemoglobinemia; May cause kidney injury; [ICSC] A skin, eye, and upper respiratory tract irritant; Ingestion of toxic doses can cause hemolysis, methemoglobinemia, cyanosis, acute renal failure, and liver injury; [HSDB] A strong oxidizing agent that reacts explosively with organic materials; An eye irritant; Ingestion can cause respiratory failure and injury to kidneys and liver; [CAMEO] Can cause combustion or explosion on contact with combustible or flammable substances; Ingestion may cause methemoglobinemia and kidney injury; [CHEMINFO] An eye and mild skin irritant; May cause skin sensitization; [eChemPortal: ERMA] An oxidizing agent; May cause explosion on contact with combustible material; An irritant; Can induce methemoglobinemia; [MSDSonline] See "CHLORATES."
Reference Link #1
Biomedical References
Exposure Assessment
Lethal Concentration
LC50 (rat) > 7,000 mg/m3/4h
Adverse Effects
Anemia
Hemolytic anemia
Methemoglobinemia
MetHgb is secondary toxic effect
Hepatotoxin
Hepatoxic (a) from occupational exposure (secondary effect) or (b) in animal studies or in humans after ingestion
Nephrotoxin
Yes
Diseases, Processes, and Activities Linked to This Agent
Processes
Industrial Processes with risk of exposure: